Overview
Dosing for RYBREVANT FASPRO™ + LAZCLUZE®
Premedications1
Administer antihistamine, antipyretic, and glucocorticoids*:
15 to 90 minutes prior to each injection according to the recommended premedications in the table below.
*Glucocorticoid administration is required for week 1, day 1 dose only and upon reinitiation after prolonged dose interruptions, then as necessary for subsequent injections.
See recommended premedicationsRapid injection(s)1†
†Administration time only; actual clinic time may vary. If multiple dosing syringes are required, administer each injection consecutively in separate quadrants of the abdomen, with each injection taking ~5 minutes.
See administration detailsProactive medications1
To help reduce the risk of select ARs
- Dermatologic AR prophylaxis:
- Oral/topical antibiotics and ceramide-based moisturizer
- Advise patients to limit direct sun exposure‡
- VTE concomitant medications: Anticoagulant prophylaxis for the first 4 months of treatment
‡During and for 2 months after treatment.
See Proactive Therapy ManagementAR, adverse reaction; VTE, venous thromboembolism.

RYBREVANT FASPRO™ Administration & Management Guide
Download a guide with comprehensive dosing, administration, and proactive therapy management details for RYBREVANT FASPRO™–based regimens.
DownloadDosing Schedule
Recommended dosing schedule for RYBREVANT FASPRO™ + LAZCLUZE® (Q4W or Q2W)
- Familiar tolerability: No new safety signals for Q4W in the Prescribing Information1,2
- Pharmacokinetics (PK) profile was consistent with Q2W dosing1-4




RYBREVANT FASPRO™
The recommended dosage of RYBREVANT FASPRO™ is based on baseline body weight and administered as a subcutaneous injection. Please read the full Prescribing Information for RYBREVANT FASPRO™ for units of hyaluronidase for each dose and for any additional dosage and administration guidance.1
Contraindications
RYBREVANT FASPRO™ is contraindicated in patients with known hypersensitivity to hyaluronidase or to any of its excipients.1
With LAZCLUZE®
Administer RYBREVANT FASPRO™ any time after LAZCLUZE® when given on the same day. Refer to the full Prescribing Information for LAZCLUZE® for recommended LAZCLUZE® dosage and administration information.1
Patients currently receiving RYBREVANT® may switch to RYBREVANT FASPRO™
If switching from RYBREVANT® Q2W dosing to RYBREVANT FASPRO™ Q4W or Q2W, switch patients at their next scheduled dose on or after week 5.1
*Following weekly doses from weeks 1 to 4.1
†Administration time only; actual clinic time may vary.1
‡Divide doses requiring greater than 15 mL into approximately equal volumes in 2 syringes. Each injection volume should not exceed 15 mL. If multiple dosing syringes are required, administer each injection consecutively in separate quadrants of the abdomen, with each injection taking ~5 minutes.1
§Dose adjustments not required for subsequent body weight changes.1
Q2W, once every 2 weeks; Q4W, once every 4 weeks.
Premedications
Premedications for RYBREVANT FASPRO™1
ARR management

*Glucocorticoid administration is also required after prolonged dose interruptions, then as necessary; or at a subsequent dose in the event of an ARR.1
Prophylactic and concomitant medications1
Dermatologic AR prophylaxis
Prophylactic measures (eg, use of oral/topical antibiotics) are recommended to reduce the risk of dermatologic ARs. When initiating treatment with RYBREVANT FASPRO™, ceramide-based moisturizer is recommended.
See additional strategies for dermatologic AR prophylaxisVTE concomitant medications
When initiating treatment with RYBREVANT FASPRO™ in combination with LAZCLUZE®, implement anticoagulant prophylaxis to reduce the risk of VTE events for the first 4 months of treatment.
- The use of vitamin K antagonists is not recommended
- If there are no signs or symptoms of VTE during the first 4 months of treatment, consider discontinuation of anticoagulant prophylaxis at the discretion of the healthcare provider
Refer to the full Prescribing Information for LAZCLUZE® for information about concomitant medications.
ARR, administration-related reaction; IV, intravenous.
Preparation
Vial selection and dose volume1
RYBREVANT FASPRO™ with LAZCLUZE® or as a single agent (Q4W or Q2W)


*Divide the 22 mL dose volume approximately equally into 2 syringes (each injection volume should not exceed 15 mL).1
†Divide the 29 mL dose volume approximately equally into 2 syringes (each injection volume should not exceed 15 mL).1
‡For the 29 mL dose volume, use one 2,240 mg amivantamab and 28,000 units hyaluronidase/14 mL vial and one 2,400 mg amivantamab and 30,000 units hyaluronidase/15 mL vial to minimize waste. If a different combination of vials is used, discard unused portion.1


Please see the Prescribing Information for RYBREVANT FASPRO™ for units of hyaluronidase for each dose.
Preparation1
STEP
1
Remove from fridge
Remove the appropriate number of RYBREVANT FASPRO™ vials from refrigerated storage
- During preparation and prior to administration, check the vial labels to ensure that the drug being prepared and administered is subcutaneous RYBREVANT FASPRO™ and not RYBREVANT®
- Do not substitute RYBREVANT FASPRO™ for or with RYBREVANT®
STEP
2
Bring to room temp

Wait at least 15 minutes to allow vial(s) to reach room temperature
- Do not warm any other way. Do not shake or dilute
STEP
3
Inspect visually

Once room temperature, inspect visually for particulate matter and discoloration prior to administration
- RYBREVANT FASPRO™ is a clear to opalescent and colorless to pale yellow solution
- Do not use if the solution is discolored or cloudy, or if foreign particles are present
STEP
4
Withdraw dose
Withdraw the required injection volume from the vial(s) into a syringe(s) using a transfer needle
- RYBREVANT FASPRO™ is compatible with stainless steel injection needles, PP and PC syringes, and PE, PU, and PVC subcutaneous infusion sets. Administer using a 21G to 23G needle or infusion set to ensure ease of administration
Vial storage1
Store RYBREVANT FASPRO™ vials in a refrigerator in original carton to protect from light. Do not freeze or shake.
Syringe storage1
- RYBREVANT FASPRO™ does not contain an antimicrobial preservative. The prepared syringes should be administered immediately
- If immediate administration is not possible, replace the transfer needle with a syringe closing cap for transport, and store the prepared syringes of RYBREVANT FASPRO™ refrigerated at 36 °F to 46 °F (2 °C to 8 °C) for up to 24 hours followed by at room temperature of 59 °F to 86 °F (15 °C to 30 °C) for up to 24 hours
- Discard the prepared syringe(s) if stored for more than 24 hours refrigerated or more than 24 hours at room temperature
- If stored in the refrigerator, allow the solution to come to room temperature before administration
PC, polycarbonate; PE, polyethylene; PP, polypropylene; PU, polyurethane; PVC, polyvinyl chloride.
Administration
Administering subcutaneous RYBREVANT FASPRO™ with LAZCLUZE®1
Administer premedications before each RYBREVANT FASPRO™ dose as recommended to reduce the risk of ARRs.
Administering RYBREVANT FASPRO™
RYBREVANT FASPRO™ is for subcutaneous use only. Do not administer RYBREVANT FASPRO™ intravenously
Once the syringe(s) is prepped, prepare to administer the injection. RYBREVANT FASPRO™ must be administered by a healthcare professional
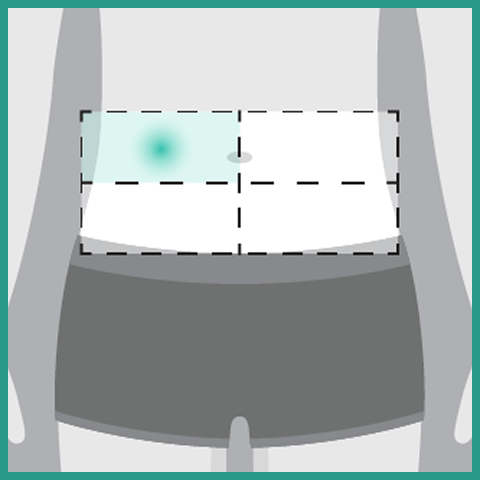
- Administer each injection of RYBREVANT FASPRO™ subcutaneously in the abdomen over approximately 5 minutes
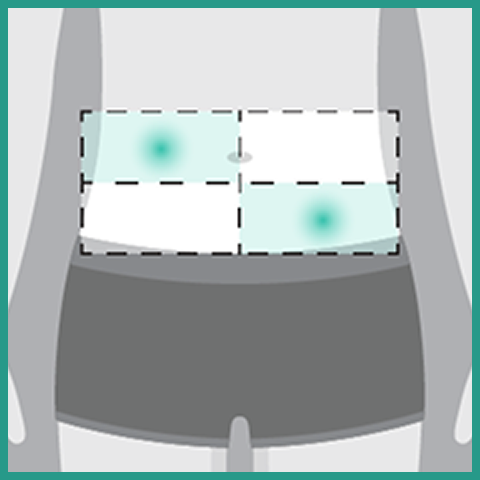
- If the total dose requires multiple injections of RYBREVANT FASPRO™, administer each injection consecutively in separate quadrants of the abdomen, with each injection taking approximately 5 minutes
- Rotate injection sites at the next scheduled dose
- Do not inject into tattoos or scars or areas where the skin is red, bruised, tender, hard, not intact, or within 2 inches (5 cm) around the periumbilical area
- If the patient experiences pain, pause or slow down delivery
- If the pain is not alleviated by pausing or slowing down the delivery rate, deliver the rest of the dose in a second injection site on the opposite side of the abdomen
- If administering with a subcutaneous infusion set, ensure the full dose is delivered through the infusion set. A 0.9% sodium chloride solution may be used to flush the remaining liquid through the line
- Discard unused portion
Administering LAZCLUZE®5
When given in combination with LAZCLUZE®, administer RYBREVANT FASPRO™ any time after LAZCLUZE® when given on the same day
- Administer LAZCLUZE® 240 mg orally once daily
- Swallow LAZCLUZE® tablets whole (with or without food). Do not crush, split, or chew
- If a patient misses a dose of LAZCLUZE® within 12 hours, instruct the patient to take the missed dose. If more than 12 hours have passed since the dose was to be given, instruct the patient to take the next dose at its scheduled time
- If vomiting occurs any time after taking LAZCLUZE®, instruct the patient to take the next dose at its next regularly scheduled time
Drug interactions with LAZCLUZE®5
Avoid concomitant use of LAZCLUZE® with strong and moderate CYP3A4 inducers. Consider an alternate concomitant medication with no potential to induce CYP3A4.
Monitor for adverse reactions associated with a CYP3A4 or BCRP substrate where minimal concentration changes may lead to serious adverse reactions, as recommended in the approved product labeling for the CYP3A4 or BCRP substrate.
Please see the full Prescribing Information for LAZCLUZE® for information regarding dosing and drug interactions.
BCRP, breast cancer resistance protein; CYP3A4, cytochrome P450 3A4.
References:
- RYBREVANT FASPRO™ [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
- Scott SC, Dias JM, Liu B, et al. PALOMA-2: subcutaneous amivantamab administered every 4 weeks plus lazertinib in first-line EGFR-mutated advanced NSCLC. Presented at: World Conference on Lung Cancer; September 6-9, 2025; Barcelona, Spain.
- Lim SM, Tan JL, Dias JM, et al. Subcutaneous amivantamab and lazertinib as first-line treatment in patients with EGFR-mutated advanced non-small cell lung cancer (NSCLC): interim results from the phase 2 PALOMA-2 study. Presented at: American Society of Clinical Oncology Congress; May 31-June 3, 2024; Chicago, IL.
- Leighl NB, Minchom AR, Lee KH, et al. Subcutaneous amivantamab administered every 4 weeks (Q4W) in patients with advanced solid malignancies: the phase 1b PALOMA study. Presented at: European Lung Cancer Congress; March 20-23, 2024; Prague, Czech Republic.
- LAZCLUZE® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.

