Overview
Use proactive therapy management to help optimize outcomes1
Studies incorporating proactive therapy management demonstrated significantly reduced rates of key ARs2-4
Choose RYBREVANT FASPRO™
80%
reduction in ARRs
with RYBREVANT FASPRO™
vs RYBREVANT® (amivantamab-vmjw)2
Rate of ARRs* (%)2
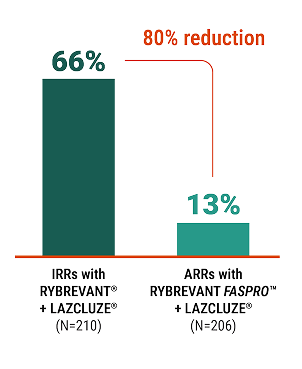

*In clinical trials of RYBREVANT® and the Prescribing Information for RYBREVANT®, the term “infusion-related reactions” was used instead of “administration-related reactions.”
Initiate prophylactic antibiotics
with skincare management
design
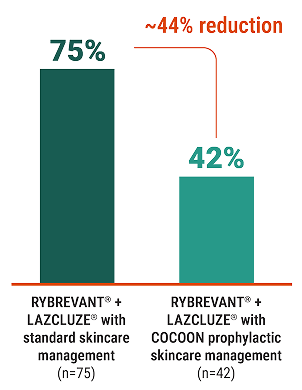
44%
reduction in grade ≥2 key dermatologic ARs
with antibiotics and skincare management3
Rate of grade ≥2 key
dermatologic ARs (%)3

Initiate anticoagulants
when combined with LAZCLUZE® (lazertinib)
7%
rate of VTE when using anticoagulants
for the first 4 months of treatment with RYBREVANT FASPRO™ + LAZCLUZE®4†
Rate of VTE (%)2,4
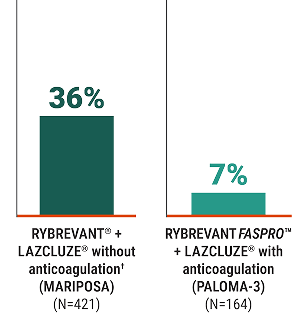

†Among all patients in PALOMA-3 receiving RYBREVANT FASPRO™ + LAZCLUZE® (N=206), VTE rate was 11%.3
‡~97% of patients in the RYBREVANT® + LAZCLUZE® arm of MARIPOSA did not receive prophylactic anticoagulation for the first 4 months.1
AR, adverse reaction; ARR, administration-related reaction; IRR, infusion-related reaction; VTE, venous thromboembolism.
Protocol
Proactive strategies that may help patients start and stay on RYBREVANT FASPRO™ + LAZCLUZE®
Premedications1
Glucocorticoid required prior to initial injection (week 1, day 1).*
Antihistamine and antipyretic required prior to each injection.
Month
1
2
3
4
5
AT-HOME ORAL AND TOPICAL REGIMEN2,3
AT-HOME ORAL AND TOPICAL REGIMEN2,3
Medications
Daily skincare
Medications
Oral doxycycline or minocycline 100 mg twice daily for first 3 months of treatment
Topical clindamycin 1% lotion on the scalp, at month 4 and for next 9 months of treatment
Oral anticoagulant for first 4 months of treatment
Daily skincare
Ceramide-based skin moisturizer
on the face and body (except scalp)
Chlorhexidine 4% wash
on the hands and feet (once daily)
Protective clothing and broad-spectrum UVA/UVB sunscreen
(SPF ≥30)
Month = month of treatment with RYBREVANT FASPRO™ and LAZCLUZE®.
*Glucocorticoid administration is also required upon reinitiation after prolonged dose interruptions, then as necessary; or at a subsequent dose in the event of an ARR.3
SPF, sun protection factor; UVA, ultraviolet A; UVB, ultraviolet B.
Expand to learn more about the impact of prophylaxis and recommended protocols
Choose RYBREVANT FASPRO™ for lower rate of ARRs
Lower rates of ARRs were observed with RYBREVANT FASPRO™ vs RYBREVANT® in the PALOMA-3 trial2
*In clinical trials of RYBREVANT® and the Prescribing Information for RYBREVANT®, the term "infusion-related reactions" was used instead of "administration-related reactions."
Premedicate with antihistamines, antipyretics, and glucocorticoids and administer RYBREVANT FASPRO™ as recommended2
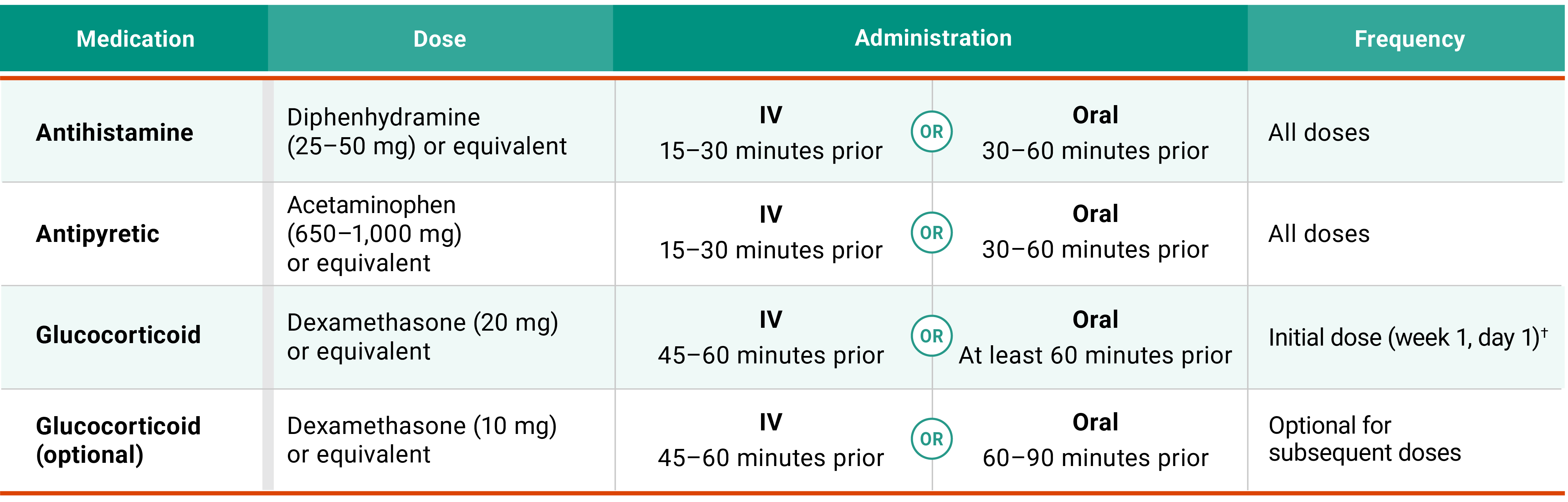

†Glucocorticoid administration is also required after prolonged dose interruptions, then as necessary for subsequent injections.
IV, intravenous.
Initiate prophylactic antibiotics with skincare management to reduce the risk of dermatologic ARs
COCOON is a phase 2, open-label, multicenter, randomized study evaluating the effect of enhanced (N=99) vs standard (N=100) dermatologic management strategies in patients treated with RYBREVANT® + LAZCLUZE® in 1L. The primary endpoint is incidence of grade ≥2 dermatologic ARs of interest in the first 12 weeks after treatment initiation.3
1L, first-line.
COCOON prophylactic skincare management includes easily accessible interventions2,3
Months 1—3

Oral antibiotic
Doxycycline or minocycline
100 mg
twice daily
Months 4—12

Topical antibiotic lotion
Topical clindamycin 1% lotion on the scalp once daily
Daily

Moisturizer
Ceramide-based moisturizer on the body and face (except scalp) at least once daily
Ceramide-based moisturizer on the body and face (except scalp) at least once daily

Limit direct exposure to sunlight
Wear protective clothing and broad-spectrum UVA/UVB sunscreen (SPF ≥30)
Wear protective clothing and broad-spectrum UVA/UVB sunscreen (SPF ≥30)

Antiseptic skin cleanser
Chlorhexidine 4% wash on the hands and feet once daily for paronychia
At the time of prespecified interim analysis, COCOON prophylactic skincare management significantly reduced the incidence of grade ≥2 dermatologic ARs vs standard skincare management3
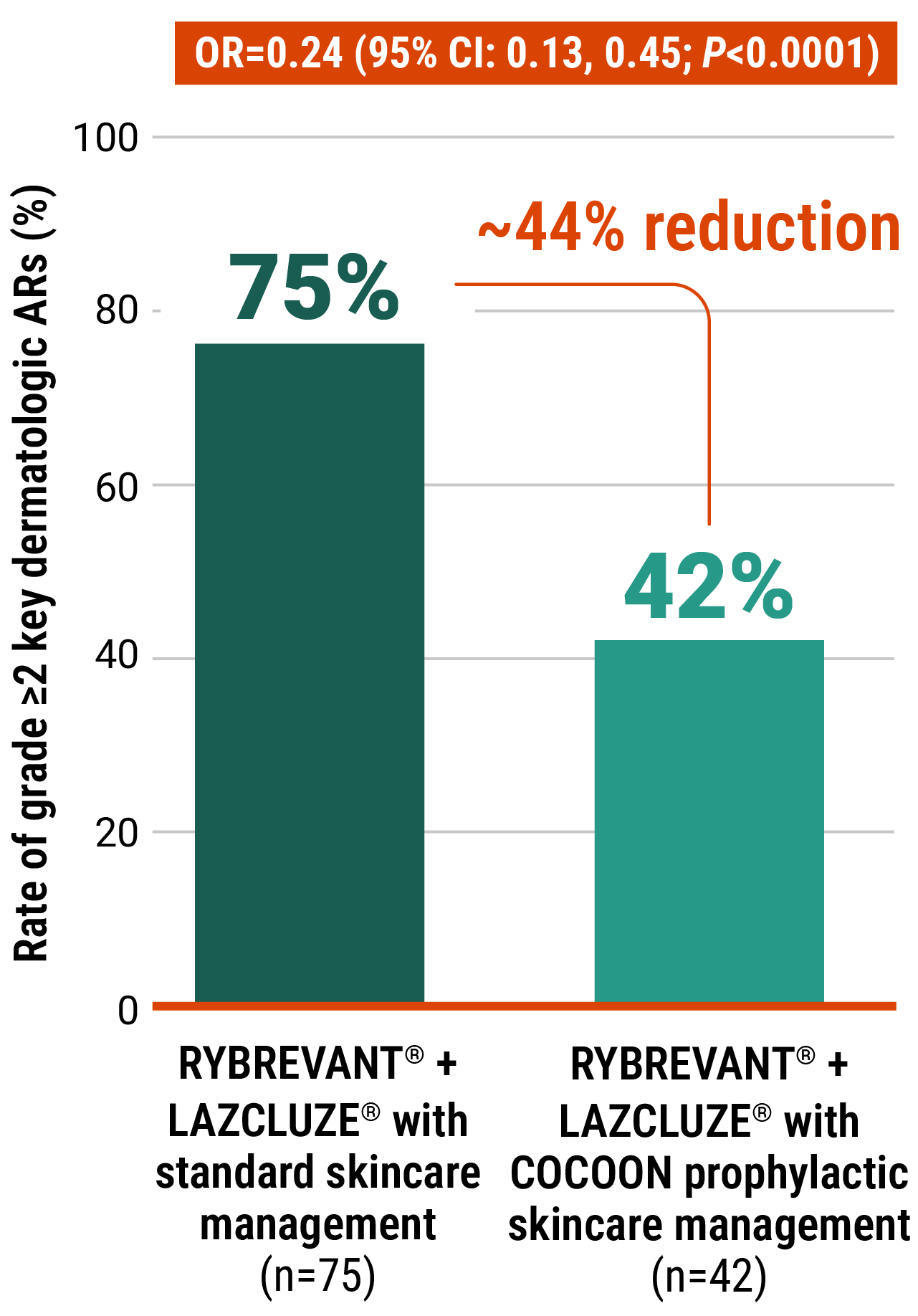
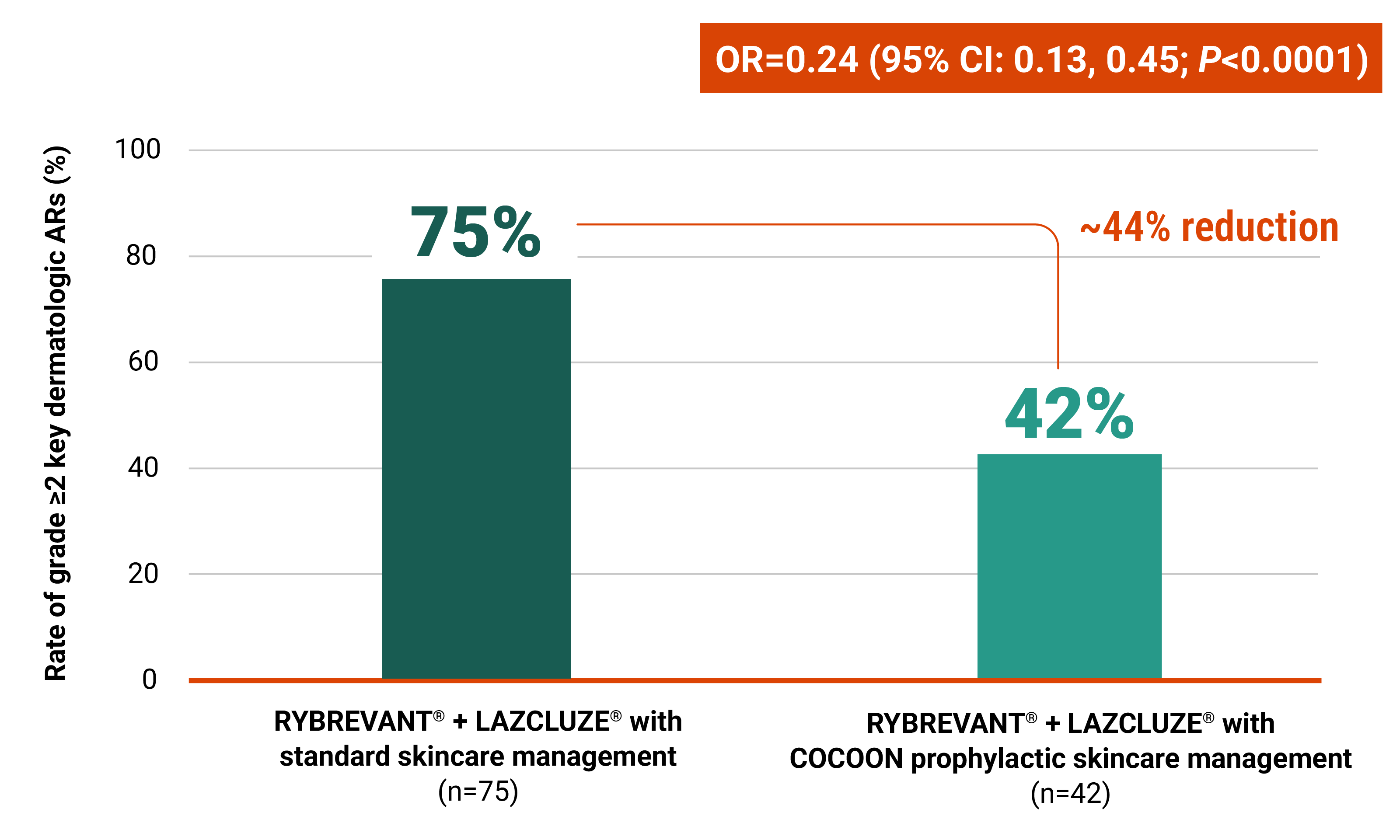
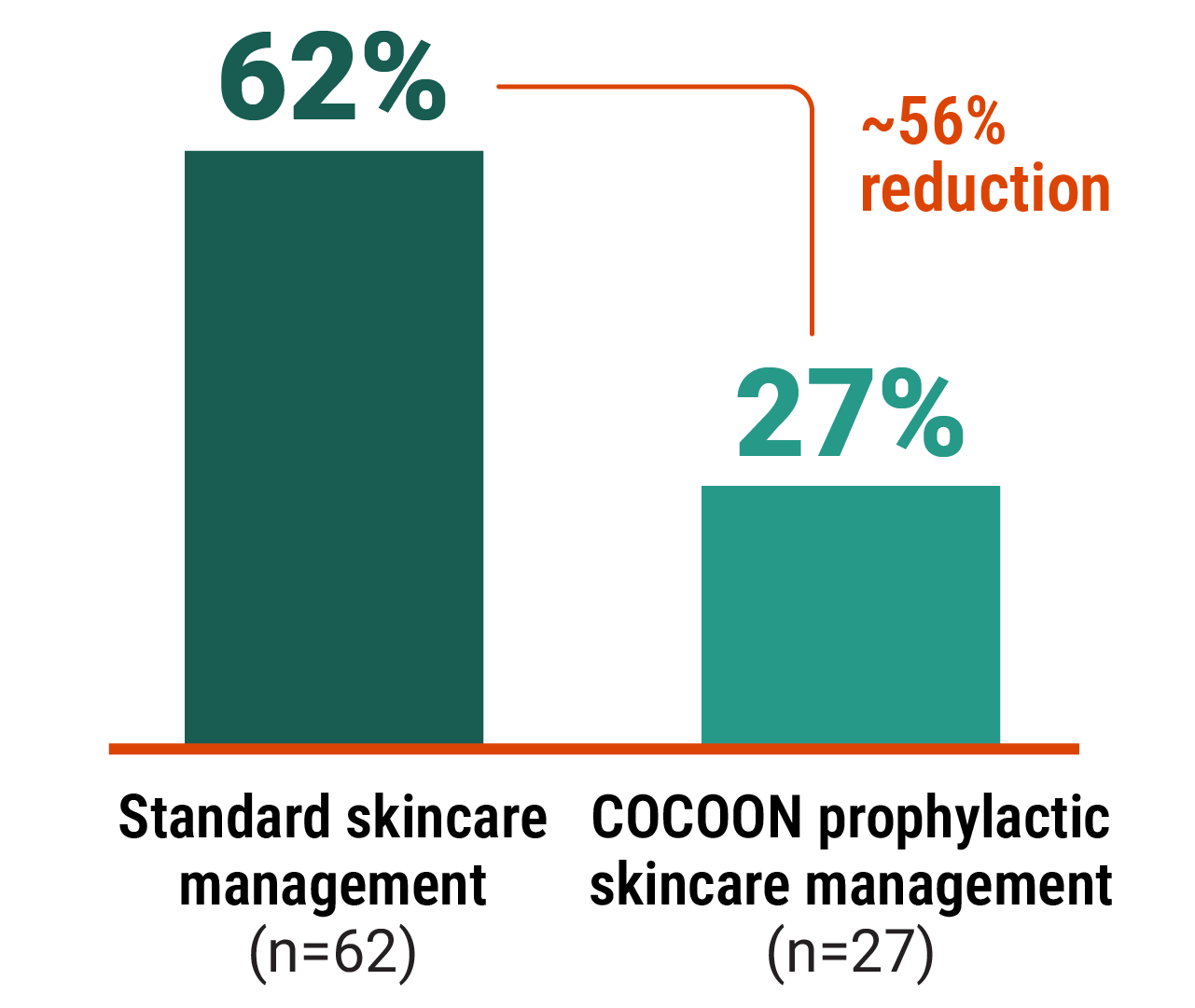
Improvements seen with the COCOON prophylactic skincare management across areas of interest3
~56% reduction in skin rash
(excluding paronychia)

>60% reduction in scalp rash
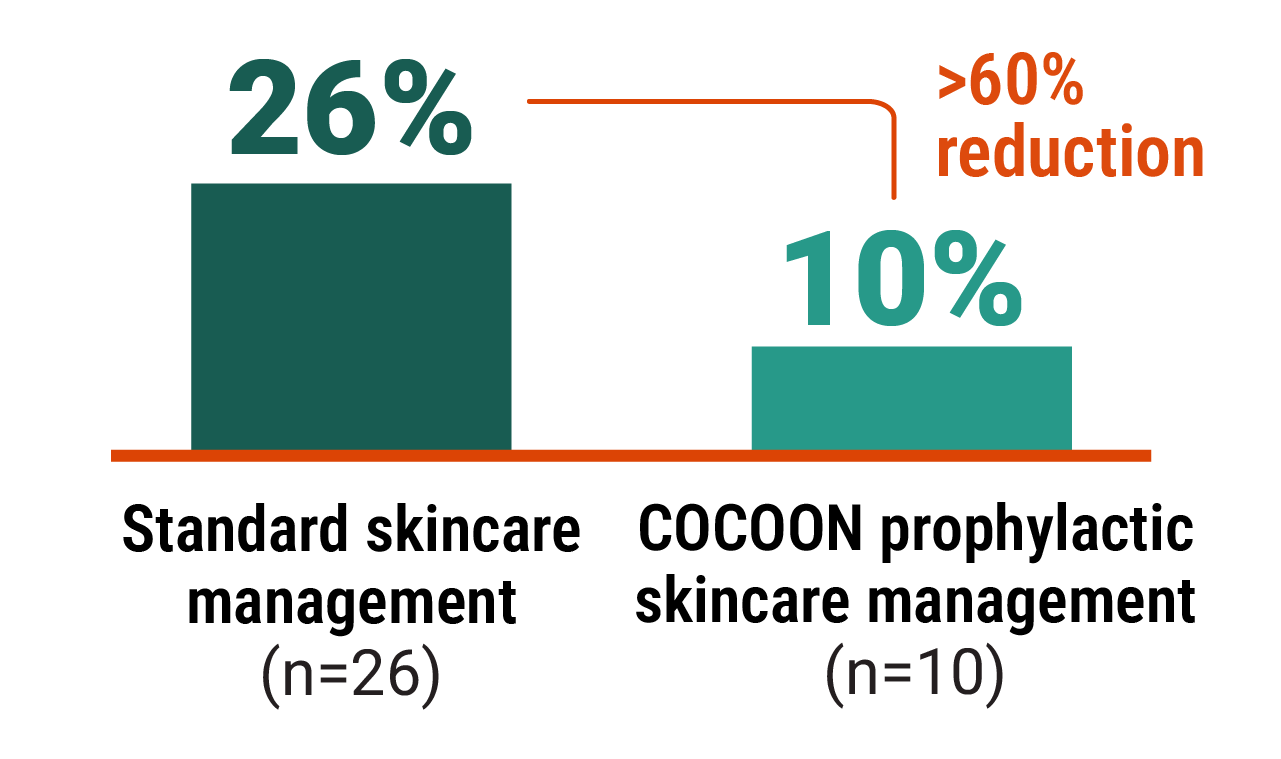
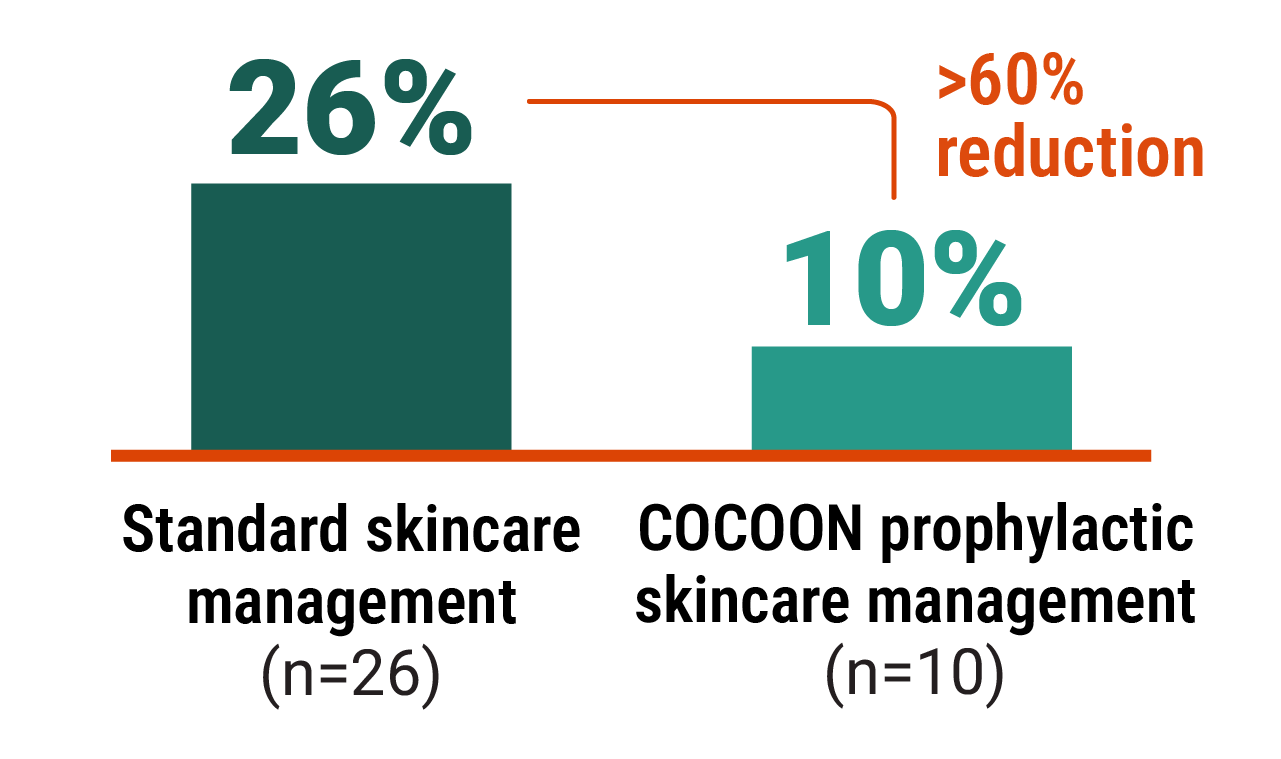
Paronychia was observed in 23% of patients receiving standard skincare management vs 21% of patients receiving COCOON prophylactic skincare management (OR=0.9 [95% CI: 0.46, 1.76]).3
CI, confidence interval; OR, odds ratio.
Initiate anticoagulants to reduce risk of VTE
80% (n=164) of patients in the RYBREVANT FASPRO™ + LAZCLUZE® arm of PALOMA-3 received prophylactic anticoagulation for the first 4 months of treatment. Among all patients receiving RYBREVANT FASPRO™ + LAZCLUZE® (N=206), VTE rate was 11%.2,4
Drug-related prophylaxis for VTE2
Prophylactic treatment with an anticoagulation medicine is recommended for the first 4 months of treatment with RYBREVANT FASPRO™ + LAZCLUZE®.
- The use of vitamin K antagonists is not recommended
- If there are no signs or symptoms of VTE during the first 4 months of treatment, consider discontinuation of anticoagulant prophylaxis at the discretion of the healthcare provider

VTE, which includes DVT and PE, is a key cause of morbidity among all patients with lung cancer7
People living with cancer have a baseline risk of developing VTE that is 9 times the risk of the general population.8
NCCN Guidelines® recommendations for cancer-associated VTE disease5
Anticoagulant options for VTE prophylaxis for ambulatory patients with cancer include direct oral anticoagulants (DOACs) and low molecular weight heparins (LMWHs).5*†‡
*Recommendations derived from clinical trials of ambulatory patients with cancer with high thrombosis risk (>18 years, Khorana VTE Risk Score of ≥2, initiating new course of chemotherapy) and are not included in product labeling. Prophylaxis duration should be 6 months or longer if risk persists.5
†Refer to the NCCN Guidelines® for the comprehensive and most up-to-date recommendations on cancer-associated VTE when considering prophylaxis.5
‡When using RYBREVANT FASPRO™ in combination with LAZCLUZE® please refer to the Prescribing Information for VTE prophylaxis recommendation.
DVT, deep vein thrombosis; PE, pulmonary embolism.
References:
- Data on file. Janssen Biotech, Inc.
- RYBREVANT FASPRO™ [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
- Cho BC, Li W, Spira AI, et al. Enhanced versus standard dermatologic management with amivantamab-lazertinib in EGFR-mutated advanced NSCLC: the COCOON global randomized controlled trial. J Thorac Oncol. 2025;20(10):1517-1530. doi:10.1016/j.jtho.2025.07.117
- Leighl NB, Akamatsu H, Lim SM, et al; PALOMA-3 Investigators. Subcutaneous versus intravenous amivantamab, both in combination with lazertinib, in refractory epidermal growth factor receptor-mutated non-small cell lung cancer: primary results from the phase III PALOMA-3 study. J Clin Oncol. 2024;42(30):3593-3605. doi:10.1200/JCO.24.01001
- Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Cancer-Associated Venous Thromboembolic Disease V.3.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed November 6, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
- Yang R, Wang H, Liu D, Li W. Incidence and risk factors of VTE in lung cancer: a meta-analysis. Ann Med. 2024;56(1):1-14. doi:10.1080/07853890.2024.2390200
- Key NS, Khorana AA, Kuderer NM, et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: ASCO Clinical Practice Guideline update. J Clin Oncol. 2020;38(5):496-520. doi:10.1200/JCO.19.01461
- Mulder FI, Horváth-Puhó E, van Es N, et al. Venous thromboembolism in cancer patients: a population-based cohort study. Blood. 2021;137(14):1959-1969. doi:10.1182/blood.2020007338

