Overview
Dosing for RYBREVANT FASPRO™ + chemotherapy
Premedications1
Administer antihistamine, antipyretic, and glucocorticoids*:
15 to 90 minutes prior to each injection according to the recommended premedications in the table below.
*Glucocorticoid administration is required for week 1, day 1 dose only and upon reinitiation after prolonged dose interruptions, then as necessary for subsequent injections.
See recommended premedicationsProactive medications1
To help reduce the risk of select ARs
- Dermatologic AR prophylaxis:
- Oral/topical antibiotics and ceramide-based moisturizer
- Advise patients to limit direct sun exposure‡
‡During and for 2 months after treatment.
See Proactive Therapy ManagementAR, adverse reaction.

RYBREVANT FASPRO™ Administration & Management Guide
Download a guide with comprehensive dosing, administration, and proactive therapy management details for RYBREVANT FASPRO™–based regimens.
DownloadDosing Schedule
Recommended dosing schedule for RYBREVANT FASPRO™ + chemotherapy (Q3W)1
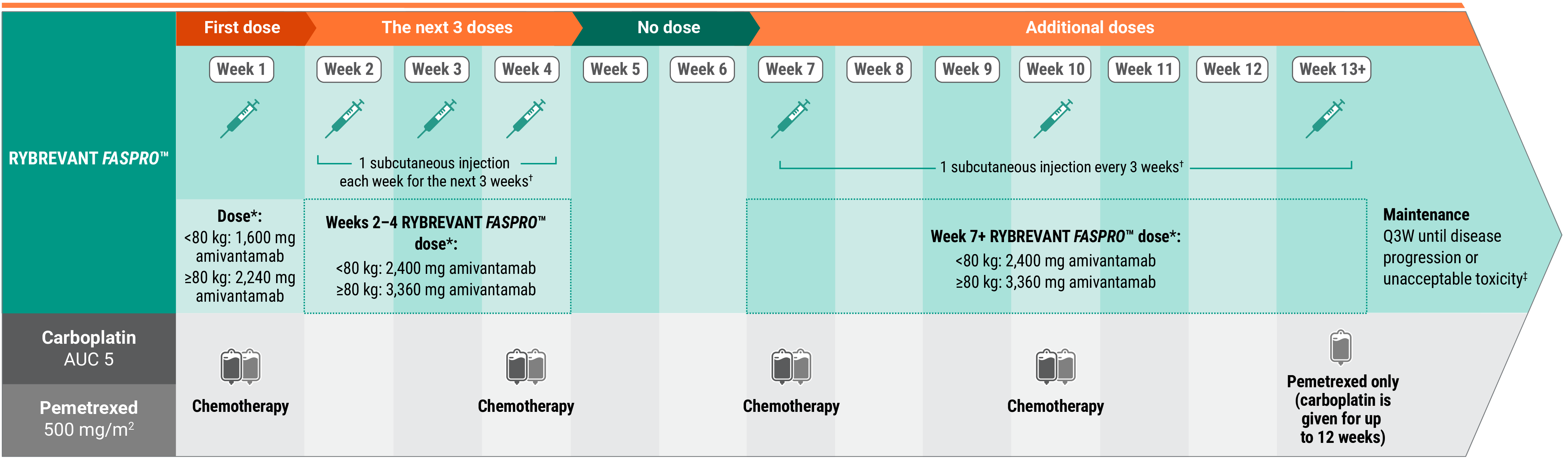
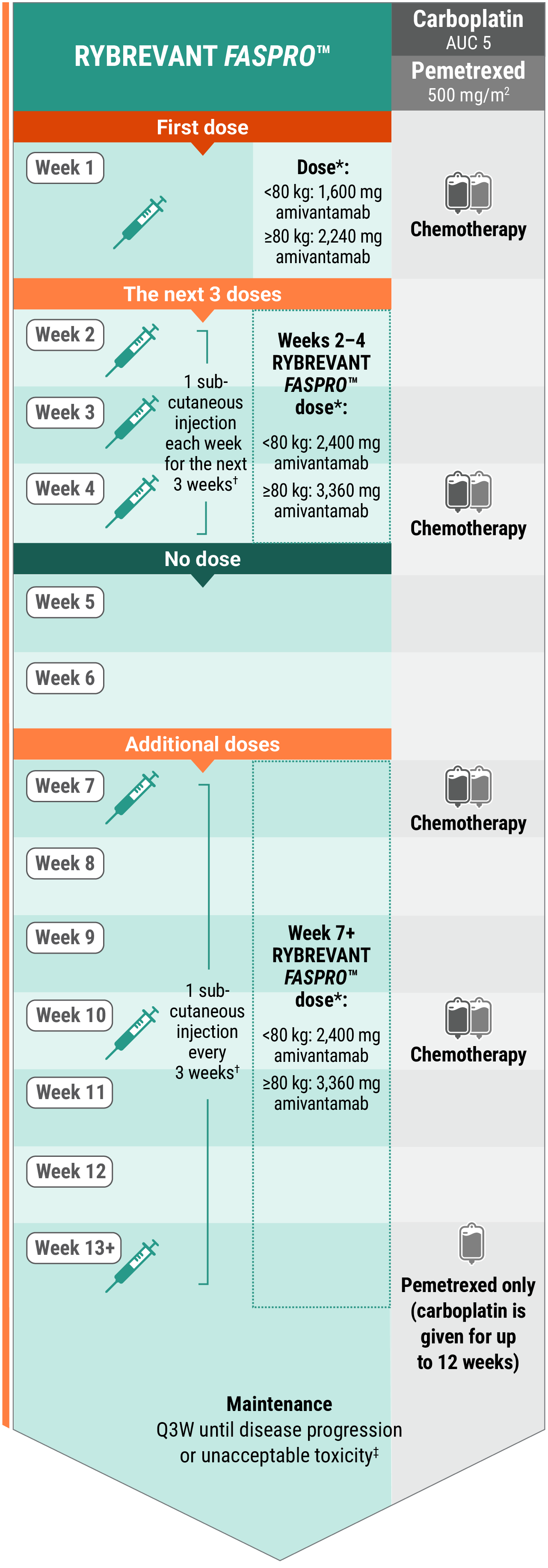
*Dose adjustments not required for subsequent body weight changes.
†Divide doses requiring greater than 15 mL into approximately equal volumes in 2 syringes and administer at separate injection sites. Each injection volume should not exceed 15 mL. Discard unused portion.
‡This refers only to RYBREVANT FASPRO™ and pemetrexed. Carboplatin should only be administered every 3 weeks for up to 12 weeks.
RYBREVANT FASPRO™
The recommended dosage of RYBREVANT FASPRO™ is based on baseline body weight and administered as a subcutaneous injection. Please see the full Prescribing Information for RYBREVANT FASPRO™ for units of hyaluronidase for each dose and for any additional dosage and administration guidance.1
Contraindications
RYBREVANT FASPRO™ is contraindicated in patients with known hypersensitivity to hyaluronidase or to any of its excipients.1
With chemotherapy
Administer RYBREVANT FASPRO™ after chemotherapy. Administer in the following order: pemetrexed, carboplatin, and then RYBREVANT FASPRO™. Refer to the full Prescribing Information for pemetrexed and carboplatin for the respective dosing information.1
If switching from RYBREVANT® (amivantamab-vmjw) Q3W dosing to RYBREVANT FASPRO™ Q3W dosing, switch patients at their next scheduled dose on or after week 41
AUC, area under the curve; Q3W, once every 3 weeks.
Premedications
Premedications for RYBREVANT FASPRO™1
ARR management
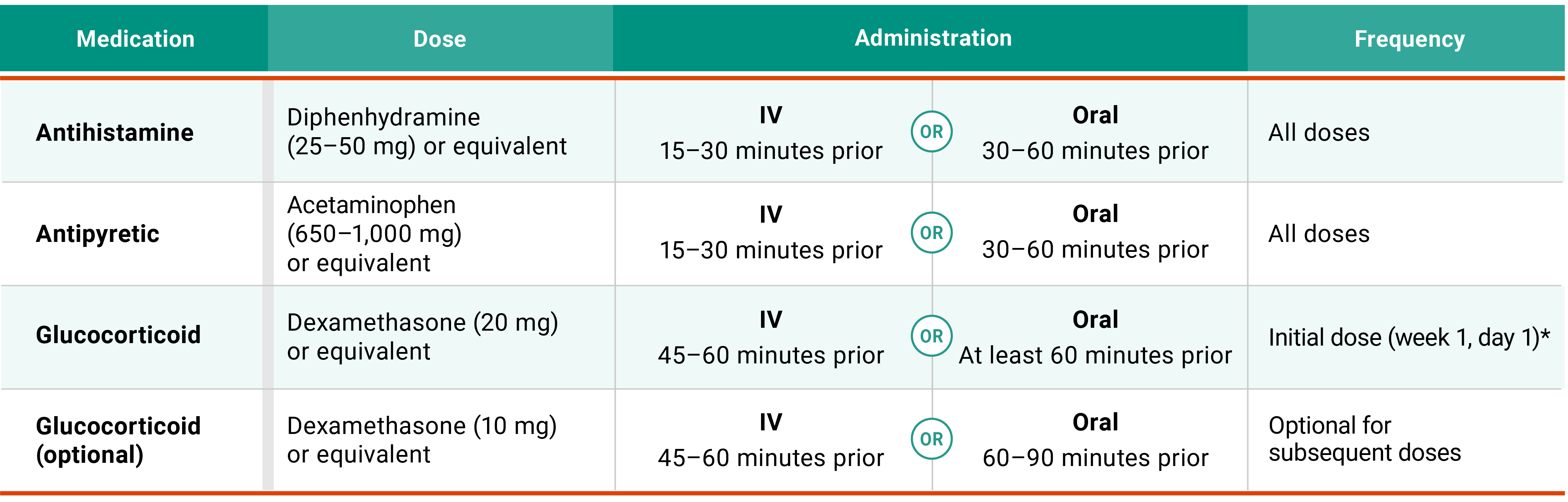

*Glucocorticoid administration is also required after prolonged dose interruptions, then as necessary for subsequent injections.
Prophylactic medications1
Dermatologic AR prophylaxis
Prophylactic measures (eg, use of oral/topical antibiotics) are recommended to reduce the risk of dermatologic ARs. When initiating treatment with RYBREVANT FASPRO™, ceramide-based moisturizer is recommended.
See additional strategies for dermatologic AR prophylaxisARR, administration-related reaction; IV, intravenous.
Preparation
Vial selection and dose volume1
RYBREVANT FASPRO™ + chemotherapy (Q3W)
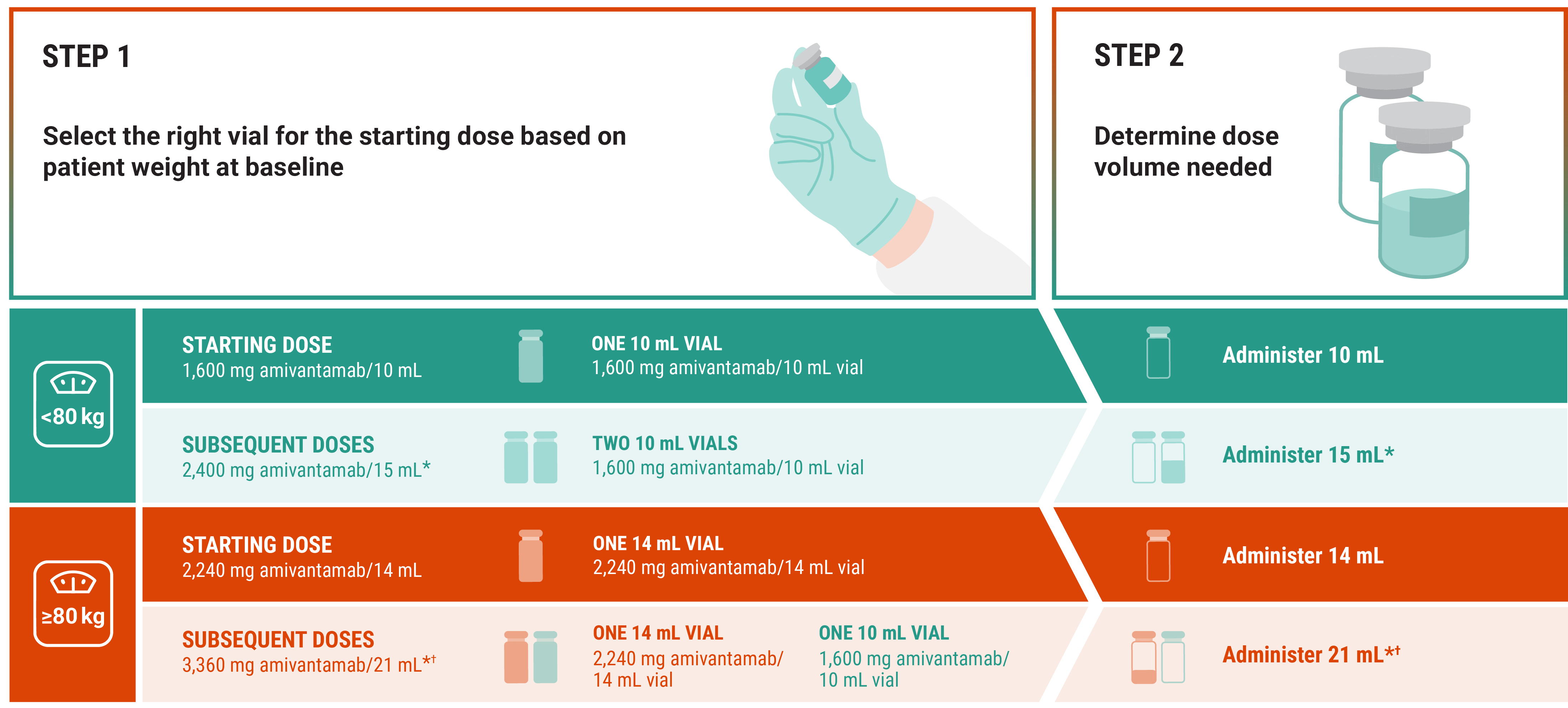
*The entire contents of all vials will not be needed. Discard unused portion.
†Divide the 21 mL dose volume approximately equally into 2 syringes (each syringe should not exceed 15 mL).
Please see the Prescribing Information for RYBREVANT FASPRO™ for units of hyaluronidase for each dose.
Preparation1
STEP
1
Remove from fridge
Remove the appropriate number of RYBREVANT FASPRO™ vials from refrigerated storage
- During preparation and prior to administration, check the vial labels to ensure that the drug being prepared and administered is subcutaneous RYBREVANT FASPRO™ and not RYBREVANT®
- Do not substitute RYBREVANT FASPRO™ for or with RYBREVANT®
STEP
2
Bring to room temp

Wait at least 15 minutes to allow vial(s) to reach room temperature
- Do not warm any other way. Do not shake or dilute
STEP
3
Inspect visually

Once room temperature, inspect visually for particulate matter and discoloration prior to administration
- RYBREVANT FASPRO™ is a clear to opalescent and colorless to pale yellow solution
- Do not use if the solution is discolored or cloudy, or if foreign particles are present
STEP
4
Withdraw dose
Withdraw the required injection volume from the vial(s) into a syringe(s) using a transfer needle
- RYBREVANT FASPRO™ is compatible with stainless steel injection needles, PP and PC syringes, and PE, PU, and PVC subcutaneous infusion sets. Administer using a 21G to 23G needle or infusion set to ensure ease of administration
- Each injection volume should not exceed 15 mL. Divide doses requiring greater than 15 mL into approximately equal volumes in 2 syringes
Vial storage1
Store RYBREVANT FASPRO™ vials in a refrigerator in original carton to protect from light. Do not freeze or shake.
Syringe storage1
- RYBREVANT FASPRO™ does not contain an antimicrobial preservative. The prepared syringes should be administered immediately
- If immediate administration is not possible, replace the transfer needle with a syringe closing cap for transport, and store the prepared syringes of RYBREVANT FASPRO™ refrigerated at 36 °F to 46 °F (2 °C to 8 °C) for up to 24 hours followed by at room temperature of 59 °F to 86 °F (15 °C to 30 °C) for up to 24 hours
- Discard the prepared syringe(s) if stored for more than 24 hours refrigerated or more than 24 hours at room temperature
- If stored in the refrigerator, allow the solution to come to room temperature before administration
PC, polycarbonate; PE, polyethylene; PP, polypropylene; PU, polyurethane; PVC, polyvinyl chloride.
Administration
Administering subcutaneous RYBREVANT FASPRO™1
Administer premedications before each RYBREVANT FASPRO™ dose as recommended to reduce the risk of ARRs.
Administering RYBREVANT FASPRO™
RYBREVANT FASPRO™ is for subcutaneous use only. Do not administer RYBREVANT FASPRO™ intravenously
Once the syringe(s) are prepped, prepare to administer the injection. RYBREVANT FASPRO™ must be administered by a healthcare professional
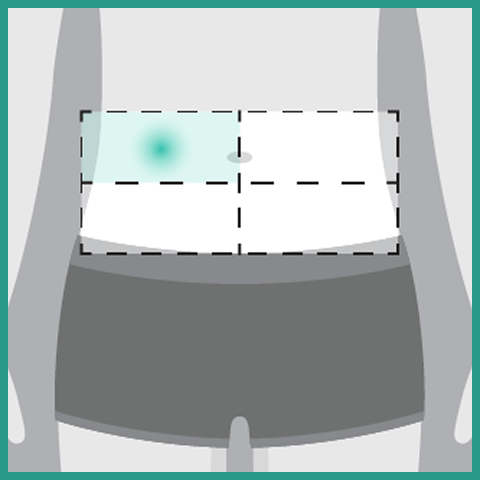
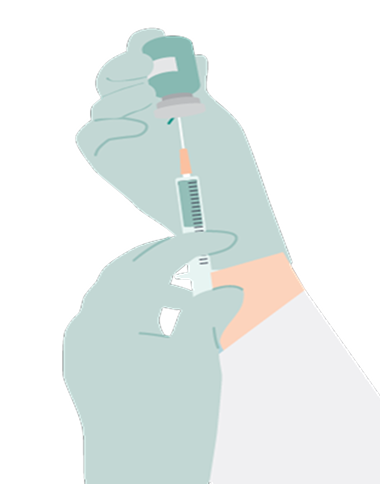
- Administer each injection of RYBREVANT FASPRO™ subcutaneously in the abdomen over approximately 5 minutes to minimize injection site irritation
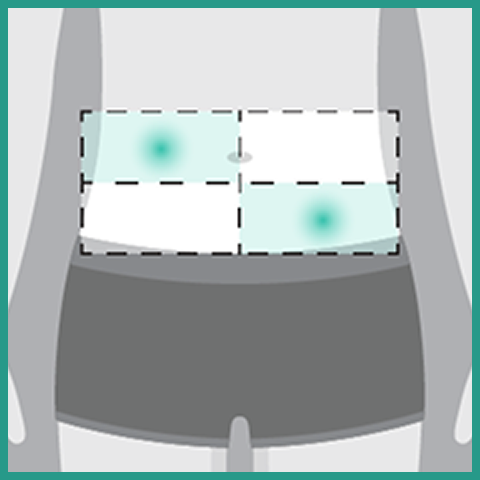
- If the total dose requires multiple injections of RYBREVANT FASPRO™, administer each injection consecutively in separate quadrants of the abdomen, with each injection taking approximately 5 minutes
- Do not inject into tattoos or scars or areas where the skin is red, bruised, tender, hard, not intact, or within 2 inches (5 cm) around the periumbilical area
- If the patient experiences pain, pause or slow down delivery
- If the pain is not alleviated by pausing or slowing down the delivery rate, deliver the rest of the dose in a second injection site on the opposite side of the abdomen
- If administering with a subcutaneous infusion set, ensure the full dose is delivered through the infusion set. A 0.9% sodium chloride solution may be used to flush the remaining liquid through the line
- Discard unused portion
Reference:
- RYBREVANT FASPRO™ [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.

