- Prescribing Information
- Important Safety Information
- Medical Information
- For US Patients
- Register for Updates
- Find a J&J Specialist
- This site is intended for
US healthcare professionals.
Navigating the Journey:
Expert Perspectives
Educational Resources About RYBREVANT FASPRO™ and RYBREVANT® (amivantamab-vmjw)-Based Regimens for EGFR-Mutated Metastatic NSCLC Care
In recent years, combination treatments for EGFR-mutated metastatic non–small cell lung cancer (NSCLC) have entered the treatment landscape, including RYBREVANT FASPRO™ + LAZCLUZE® (lazertinib) and other RYBREVANT®-based regimens.1,2 Explore this page for more information on why you and your patients should consider RYBREVANT FASPRO™ + LAZCLUZE®, with access to
- An interactive video reviewing critical unmet needs
- Expert-led content around key topics impacting patient care, including data presentations, experience perspectives, and management strategies to support ongoing adherence
[COMING SOON]
Now Available!
Dr Pradnya Patil highlights results from PALOMA-3, a randomized controlled study that compared RYBREVANT FASPRO™ with RYBREVANT®, both in combination with LAZCLUZE®.1
Watch Now
EGFR, epidermal growth factor receptor.


The Clinical Journey
The treatment landscape for EGFR-mutated NSCLC has undergone a dramatic evolution over the last 2 decades, starting with the discovery of activating mutations in the EGFR gene in 2004, which ushered in the era of targeted therapy, and has led to combination regimens.2-5
But EGFR+ metastatic NSCLC has been relentless, and <20% of patients are still alive after 5 years.2,6
Explore this interactive video to learn more about the evolution of EGFR-targeted treatments. Click to begin.




Expert Video Library
Hear oncology experts share their insights and experiences in managing EGFR+ locally advanced or metastatic NSCLC with RYBREVANT FASPRO™ and RYBREVANT®-based regimens. Click on the titles or thumbnails to watch each video and learn more about key topics impacting patient care.


A First-Line Treatment Option for EGFR+* Locally Advanced or Metastatic NSCLC
Martin Dietrich, MD, of The US Oncology and Cancer Care Centers of Brevard in Orlando, Florida, presents the results from the MARIPOSA trial, including the OS, intracranial PFS, and duration of response data.1,7
[COMING SOON]
RYBREVANT FASPRO™ + LAZCLUZE®: Redefining Care for EGFR+* Locally Advanced or Metastatic NSCLC
Pradnya Patil, MD, of The Dyson Center for Cancer Care in Poughkeepsie, New York, highlights the results from PALOMA-3, a randomized controlled study that compared RYBREVANT FASPRO™ with RYBREVANT®.1


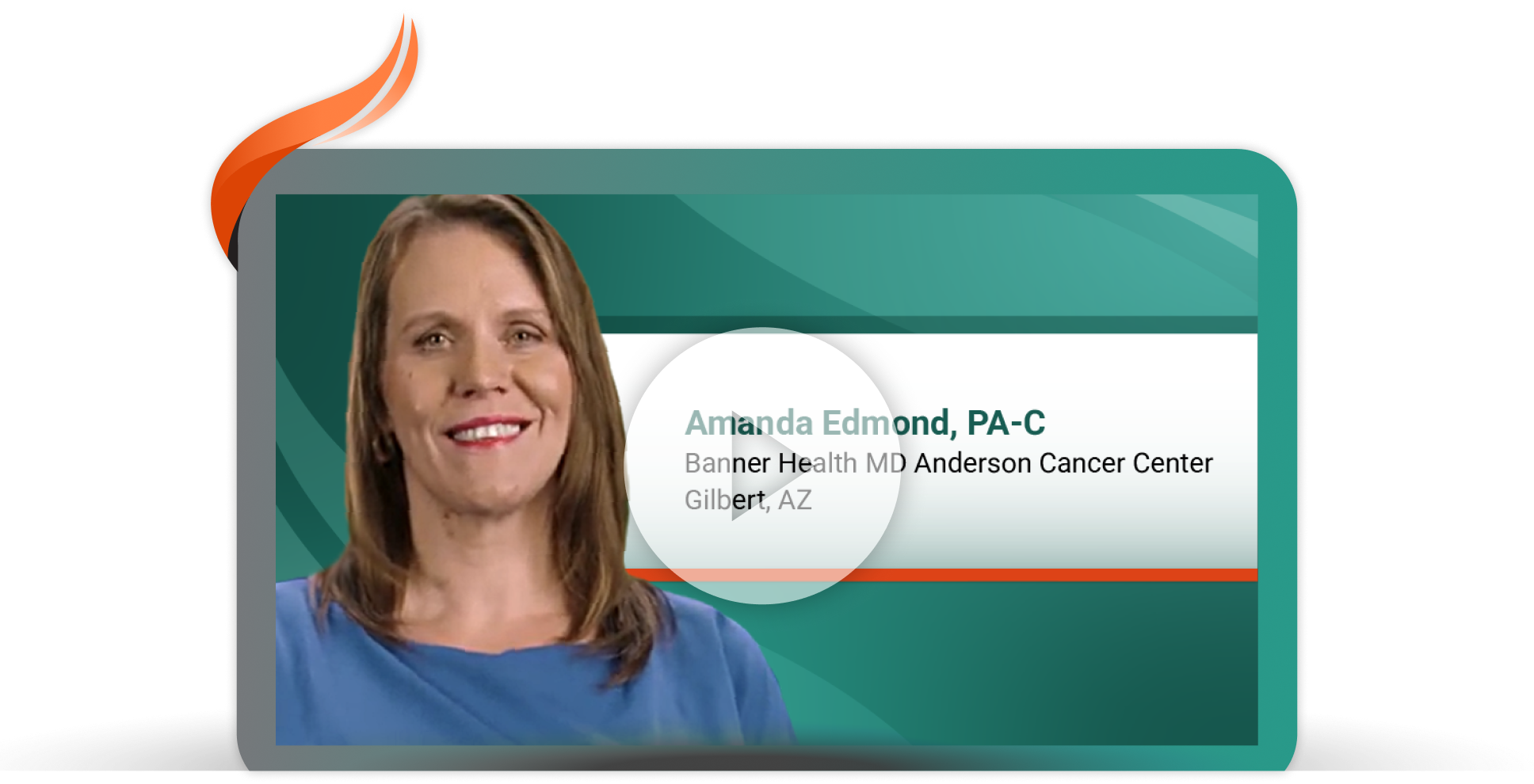

Proactive Management of Dermatologic Adverse Reactions for a First-Line Treatment Option for EGFR+* Locally Advanced or Metastatic NSCLC
Amanda Edmond, PA-C, at Banner Health MD Anderson Cancer Center in Gilbert, Arizona, shares results from COCOON, an ongoing phase 2, open-label, randomized study evaluating the effect of enhanced vs standard dermatologic management strategies in patients treated with RYBREVANT® + LAZCLUZE® in 1L.8
*Ex19del/L858R.
1L, first line; ex19del, exon 19 deletions; L858R, exon 21 L858R mutations; OS, overall survival; PFS, progression-free survival.
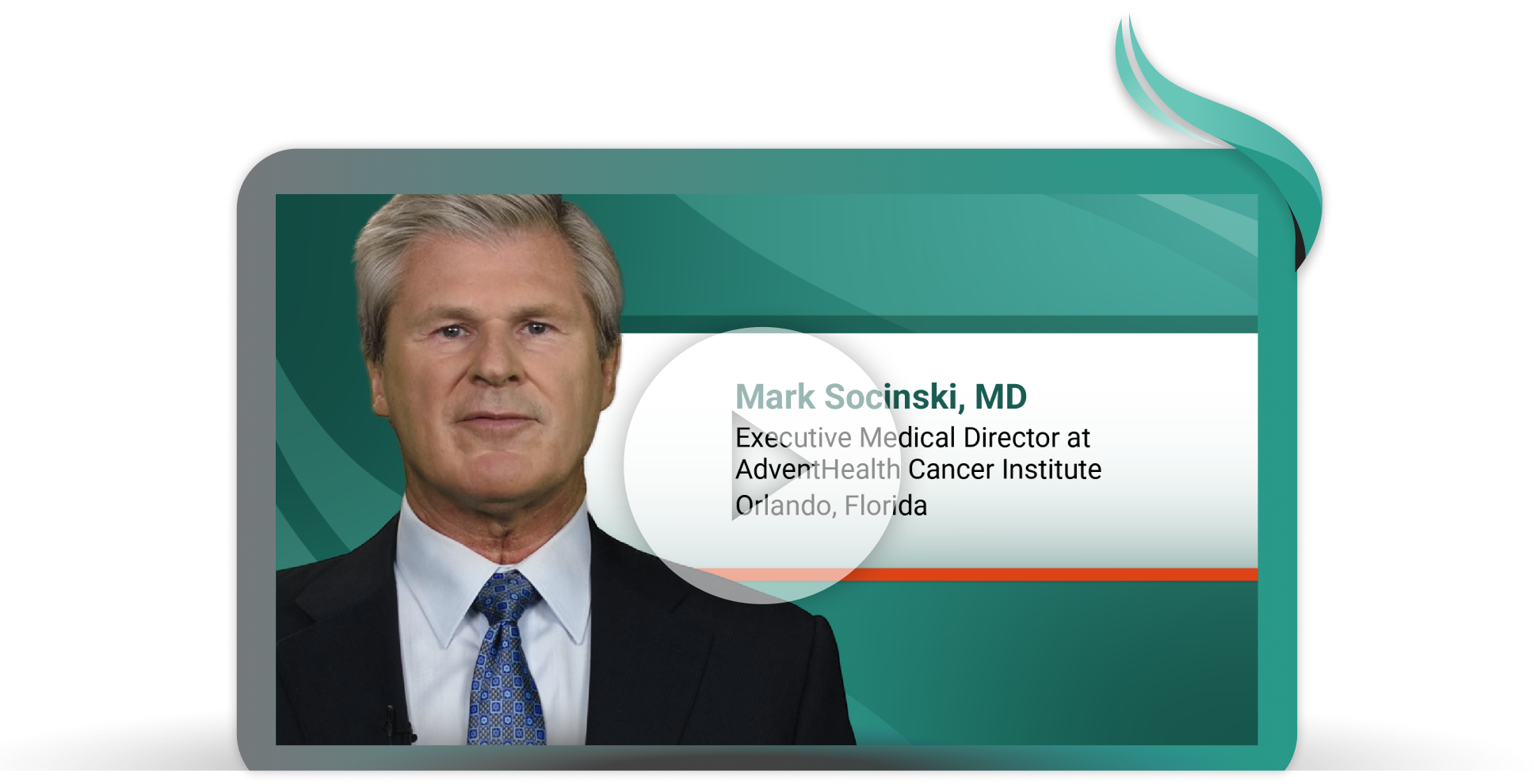
A First-Line Treatment Option for Patients With Locally Advanced or Metastatic Non-Small Cell Lung Cancer With EGFR Exon 20 Insertion Mutations
Mark Socinski, MD, executive medical director at AdventHealth Cancer Institute, shares key results from PAPILLON, a study evaluating the combination of RYBREVANT® (amivantamab-vmjw) + carboplatin + pemetrexed in patients with locally advanced or metastatic NSCLC and EGFR exon 20 insertion mutations.9

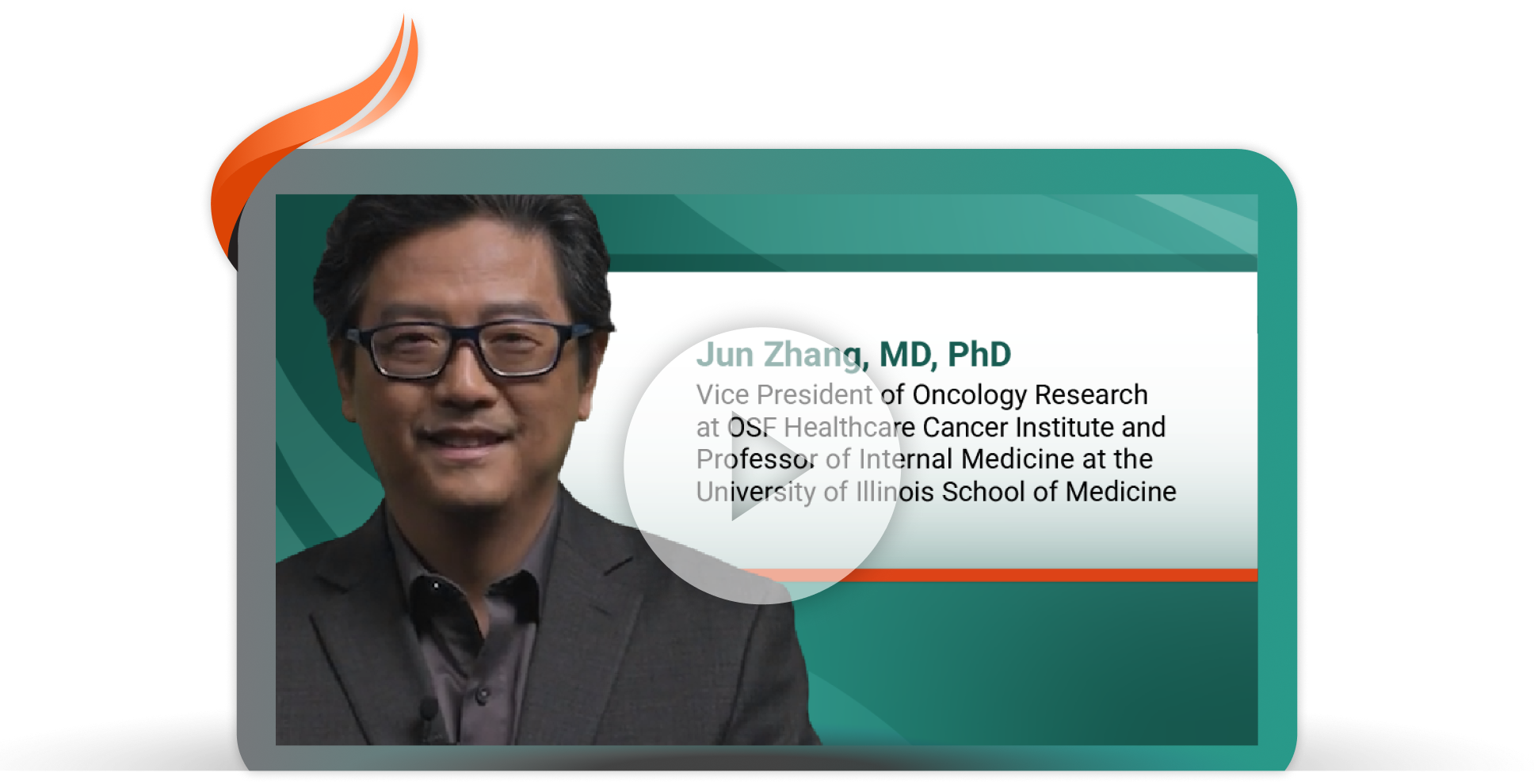

Proactive Management of Infusion-Related Reactions With a First-Line Treatment Option for EGFR+* Locally Advanced or Metastatic NSCLC†
Jun Zhang, MD, Vice President of Oncology Research at OSF Healthcare Cancer Institute and Professor of Internal Medicine at the University of Illinois School of Medicine, discusses the findings from SKIPPirr, a study that assessed prophylactic strategies to reduce incidence and/or severity of first-dose infusion-related reactions in patients on RYBREVANT® + LAZCLUZE®.10
*Ex19del/L858R.
†For RYBREVANT®-based regimens only.







In Your Practice
Managing EGFR+ NSCLC is a complex journey, and understanding the unmet needs in the treatment landscape, as well as the opportunities to address them, can help both you and your patients with 1L treatment and throughout the course of care.
TapClick on the icons for more information.
25% to 40% of patients on EGFR TKIs never receive 2L therapy11-13*
Resistance to EGFR TKIs in the current treatment landscape is inevitable2†
See MARIPOSA results, which included OS and intracranial PFS1,7
Proactive therapy management of ARs was evaluated in dedicated clinical studies8,10
For further information about RYBREVANT FASPRO™ and RYBREVANT®-based regimens, talk with a J&J specialist
*Range includes patients who died or discontinued the assigned therapy without receiving 2L therapy during follow-up.11-13
†EGFR mutations and MET amplification are the most common mechanisms of resistance among patients taking EGFR TKIs.15-17
2L, second-line; AR, adverse reaction; MET, mesenchymal-epithelial transition; TKI, tyrosine kinase inhibitor.
References:
- RYBREVANT FASPRO™ [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
- Zhou F, Guo H, Xia Y, et al. The changing treatment landscape of EGFR-mutant non-small-cell lung cancer. Nat Rev Clin Oncol. 2025;22(2):95-116. doi:10.1038/s41571-024-00971-2
- US Food and Drug Administration. Accessed October 20, 2025. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-datopotamab-deruxtecan-dlnk-egfr-mutated-non-small-cell-lung-cancer
- Lynch TJ, Bell DW, Sordella R, et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N Engl J Med. 2004;350(21):2129-2139. doi:10.1056/NEJMoa040938
- Paez JG, Jänne PA, Lee JC, et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science. 2004;304(5676):1497-1500. doi:10.1126/science.1099314
- Bazhenova L, Minchom A, Viteri S, et al. Comparative clinical outcomes for patients with advanced NSCLC harboring EGFR exon 20 insertion mutations and common EGFR mutations. Lung Cancer. 2021;162:154-161. doi:10.1016/j.lungcan.2021.10.020
- Yang JC, Lu S, Hayashi H, et al; MARIPOSA Investigators. Overall survival with amivantamab-lazertinib in EGFR-mutated advanced NSCLC. N Engl J Med. 2025. doi:10.1056/NEJMoa2503001
- Cho BC, Li W, Spira AI, et al. Enhanced versus standard dermatologic management with amivantamab-lazertinib in EGFR-mutated advanced NSCLC: the COCOON global randomized controlled trial. J Thorac Oncol. 2025:S1556-0864(25)00939-6. doi:10.1016/j.jtho.2025.07.117
- Zhou C, Tang K-J, Cho BC, et al; PAPILLON investigators. Amivantamab plus chemotherapy in NSCLC with EGFR exon 20 insertions. N Engl J Med. 2023;389(22):2039-2051. doi:10.1056/NEJMoa2306441
- Spira AI, Paz-Ares L, Han JY, et al. Preventing infusion-related reactions with intravenous amivantamab–results from SKIPPirr, a phase 2 study: a brief report. J Thorac Oncol. 2025;20(6):809-816. doi:10.1016/j.jtho.2025.01.018
- Nieva J, Karia PS, Okhuoya P, et al. A real-world (rw) observational study of long-term survival (LTS) and treatment patterns after first-line (1L) osimertinib in patients (pts) with epidermal growth factor receptor (EGFR) mutation-positive (m) advanced non-small cell lung cancer [ESMO abstract 1344P]. Ann Oncol. 2023;34(suppl 2):S774.
- Lee JY, Mai V, Garcia M, et al. Treatment patterns and outcomes of first-line osimertinib-treated advanced EGFR mutated NSCLC patients: a real-world study [IASLC abstract EP08.02-082]. Presented at: IASLC 2022 World Conference on Lung Cancer; August 6-9, 2022; Vienna, Austria.
- Girard N, Leighl NB, Ohe Y, et al. Mortality among EGFR-mutated advanced NSCLC patients after starting frontline osimertinib treatment: a real-world, US attrition analysis. Presented at: the European Lung Cancer Congress; March 29-April 1, 2023; Copenhagen, Denmark. Poster 19P.
- Hayashi H, Besse B, LS-H, et al. Mechanisms of acquired resistance to first-line amivantamab plus lazertinib vs osimertinib: updated analysis from MARIPOSA. Presented at: IASLC 2025 World Conference on Lung Cancer; September 6-9, 2025; Barcelona, Spain.
- Passaro A, Jänne PA, Mok T, Peters S. Overcoming therapy resistance in EGFR-mutant lung cancer. Nat Cancer. 2021;2(4):377-391. doi:10.1038/s43018-021-00195-8
- Fu K, Xie F, Wang F, Fu L. Therapeutic strategies for EGFR-mutated non-small cell lung cancer patients with osimertinib resistance. J Hematol Oncol. 2022;15(1):173. doi:10.1186/s13045-022-01391-4
- Yu HA, Kerr K, Rolfo CD, et al. Detection of MET amplification (METamp) in patients with EGFR mutant (m) NSCLC after first-line (1L) osimertinib. J Clin Oncol. 2023;41(16 suppl):9074.
