Overview
Guide patients toward their goals with their optimal dose1,2
INITIATE
prophylactic strategies
PAUSE
REDUCE
REEVALUATE
Based on severity and/or recurrence, certain ARs require discontinuation.1 See specific guidance for dose modifications in the section below.
Note: If a dose modification is needed for RYBREVANT®, treatment may be continued with LAZCLUZE® (lazertinib).1,2
AR, adverse reaction.
Dosage Reductions
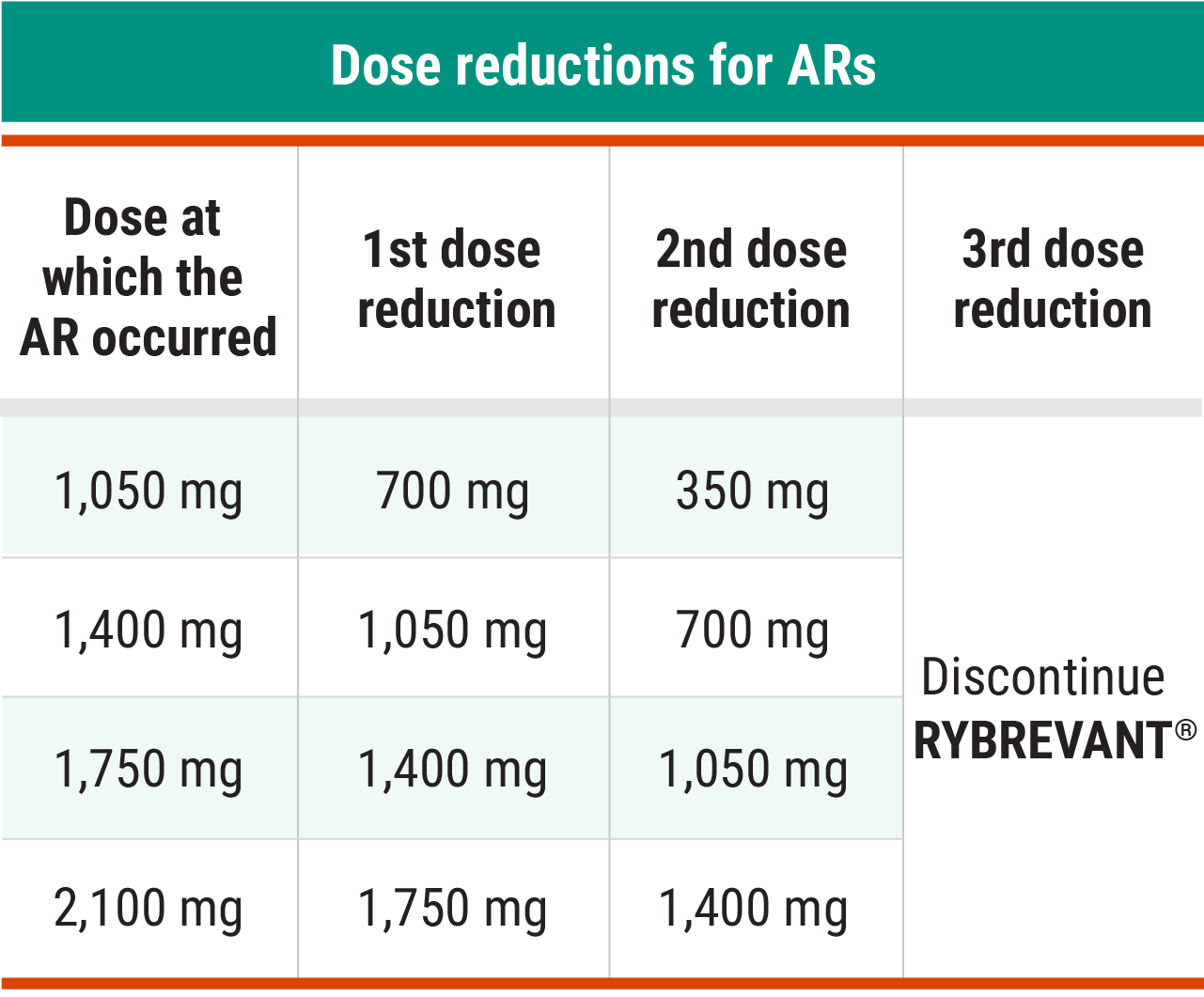
Recommended RYBREVANT® dose reductions for ARs1

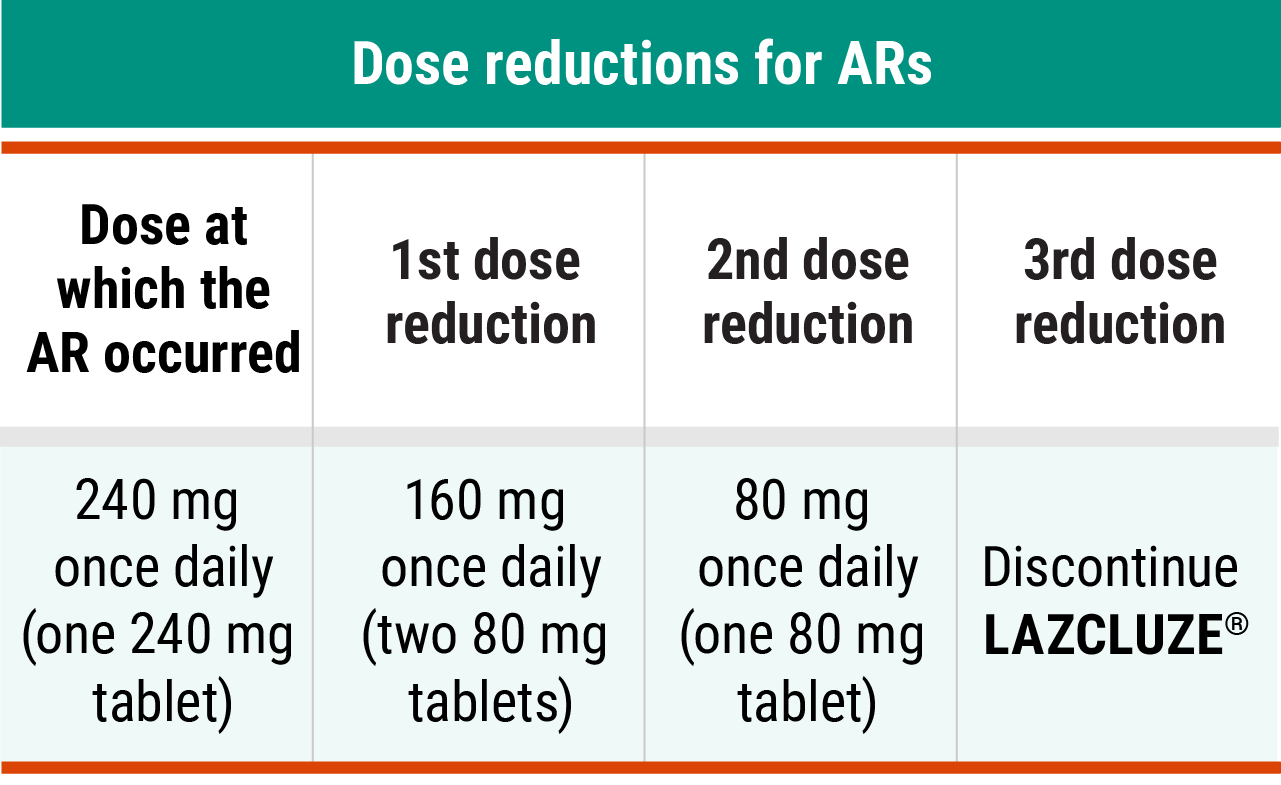
Recommended LAZCLUZE® dose reductions for ARs2
Recommended LAZCLUZE® dose reductions for ARs2

Adverse event severity scale3
Based on the Common Terminology Criteria for Adverse Events (CTCAE) v5.0*


CTCAE definition may differ from the Prescribing Information.
Instrumental ADL refer to preparing meals, shopping for groceries or clothes, using the telephone, managing money, etc.
Self-care ADL refer to bathing, dressing and undressing, feeding self, using the toilet, taking medications, and not being bedridden.
Dosage Modifications
Recommended dosage modifications for ARs1,2
Dosage modifications and management of ARs for RYBREVANT® + LAZCLUZE®
For RYBREVANT® + LAZCLUZE®, refer to both the RYBREVANT® and LAZCLUZE® recommendations.
For RYBREVANT® + LAZCLUZE®, refer to both the RYBREVANT® and LAZCLUZE® recommendations.
For RYBREVANT® + chemotherapy or RYBREVANT® as a single agent, refer only to the RYBREVANT® recommendations.
| Severity | Dosage modifications | ||
|---|---|---|---|
| Grade 2 or 3 | WithholdWithhold both drugs | AdministerAdminister anticoagulant treatment as clinically indicated | ResumeOnce anticoagulant treatment has been initiated, resume both drugs at the same dose level, at the discretion of the treating physician |
| Grade 4 or recurrent grade 2 or 3 despite therapeutic level anticoagulation | Discontinue Discontinue RYBREVANT® permanentlyWithhold Withhold LAZCLUZE® | AdministerAdminister anticoagulant treatment as clinically indicated | ResumeOnce anticoagulant treatment has been initiated, treatment can continue with LAZCLUZE® at the same dose level, at the discretion of the treating physician |
| Severity | Dosage modifications | ||
|---|---|---|---|
| Grade 1 or 2 | InterruptInterrupt RYBREVANT® infusion if suspected and monitor patient until symptoms resolve | Resume Resume the infusion at 50% of the infusion rate at which the reaction occurredIf there are no additional symptoms after 30 minutes, the infusion rate may be escalated | Include prophylaxisInclude corticosteroid with premedications for subsequent dose of RYBREVANT® |
| Grade 3 | InterruptInterrupt RYBREVANT® infusion and administer supportive care medications. Continuously monitor patient until reaction symptoms resolve | Resume Resume the infusion at 50% of the infusion rate at which the reaction occurredIf there are no additional symptoms after 30 minutes, the infusion rate may be escalated | Include prophylaxis Include corticosteroid with premedications for subsequent dose.Discontinue For recurrent grade 3, discontinue RYBREVANT® permanently |
| Grade 4(or any grade anaphylaxis/ anaphylactic reactions) | DiscontinueDiscontinue RYBREVANT® permanently |
| Severity | Dosage modifications | |
|---|---|---|
| Any grade | WithholdWithhold both drugs if suspected | DiscontinueDiscontinue both drugs permanently if confirmed |
(including dermatitis acneiform, pruritus, dry skin)
| Severity | Dosage modifications | |||
|---|---|---|---|---|
| Grade 1 | InitiateInitiate supportive care management as clinically indicated | ReassessReassess after 2 weeks; if rash does not improve, consider dose reduction of RYBREVANT® | ||
| Grade 2 | InitiateInitiate supportive care management as clinically indicated | ReassessReassess after 2 weeks; if rash does not improve, reduce RYBREVANT® dose and continue LAZCLUZE® at the same dose | ReassessReassess every 2 weeks; if no improvement, reduce LAZCLUZE® dose until grade ≤1, then may resume previous dose of LAZCLUZE® at the discretion of the healthcare provider | |
| Grade 3 | WithholdWithhold both drugs and initiate supportive care management as clinically indicated | ReassessUpon recovery to grade ≤2, resume RYBREVANT® at a reduced dose; resume LAZCLUZE® at the same dose or consider dose reduction | DiscontinueIf there is no improvement within 2 weeks, discontinue both drugs permanently | |
| Grade 4(including severe bullous, blistering, or exfoliating skin conditions, including TEN for RYBREVANT®) | DiscontinueDiscontinue RYBREVANT® permanently | InitiateInitiate supportive care management as clinically indicated | WithholdWithhold LAZCLUZE® until recovery to grade ≤2 or baseline | ResumeUpon recovery to grade ≤2, resume LAZCLUZE® at a reduced dose at the discretion of the healthcare provider |
| Severity | Dosage modifications | ||
|---|---|---|---|
| Grade 3 | WithholdWithhold both drugs until recovery to grade ≤1 or baseline | Resume Resume both drugs at the same dose if recovery occurs within 1 weekResume both drugs at reduced dose or LAZCLUZE® alone if recovery occurs after 1 week but within 4 weeks | DiscontinueDiscontinue both drugs permanently if recovery does not occur within 4 weeks |
| Grade 4 | WithholdWithhold both drugs until recovery to grade ≤1 or baseline | ResumeResume both drugs at reduced dose or LAZCLUZE® alone if recovery occurs within 4 weeks | Discontinue Discontinue both drugs permanently if recovery does not occur within 4 weeksDiscontinue RYBREVANT® permanently for recurrent grade 4 reactions |
Recommended dosage modifications for ARs for RYBREVANT® in combination with LAZCLUZE®1
When administering RYBREVANT® in combination with LAZCLUZE®, if there is an AR requiring dose reduction after withholding treatment and resolution, reduce the dose of RYBREVANT® first.
Recommended dosage modifications for ARs for RYBREVANT® in combination with carboplatin and pemetrexed1
When administering RYBREVANT® in combination with carboplatin and pemetrexed, modify the dosage of one or more drugs. Withhold or discontinue RYBREVANT® as shown in the tables above. Refer to the Prescribing Information for carboplatin and pemetrexed for additional dosage modification information.
ILD, interstitial lung disease; TEN, toxic epidermal necrolysis; VTE, venous thromboembolism.
References:
- RYBREVANT® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
- LAZCLUZE® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
- US Department of Health and Human Services. National Cancer Institute. Common terminology criteria for adverse events (CTCAE). Version 5.0. Published November 27, 2017. Accessed December 10, 2025. https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events/ctcae-v5-5x7.pdf


