EGFR+
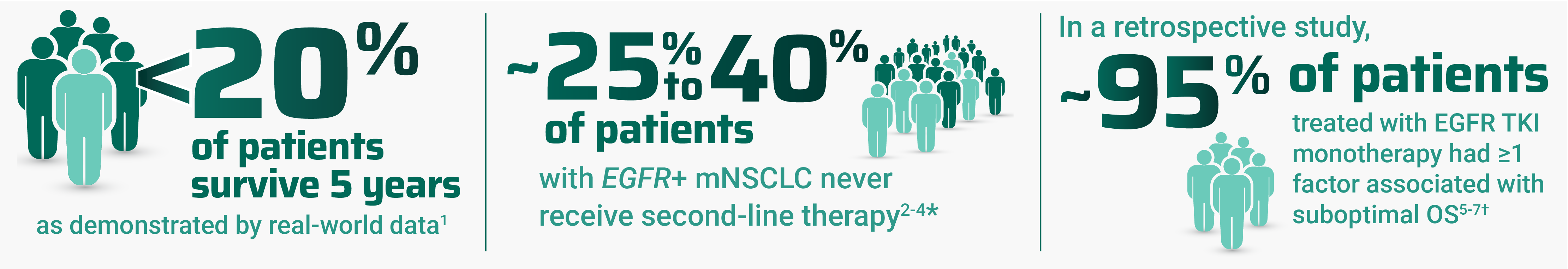
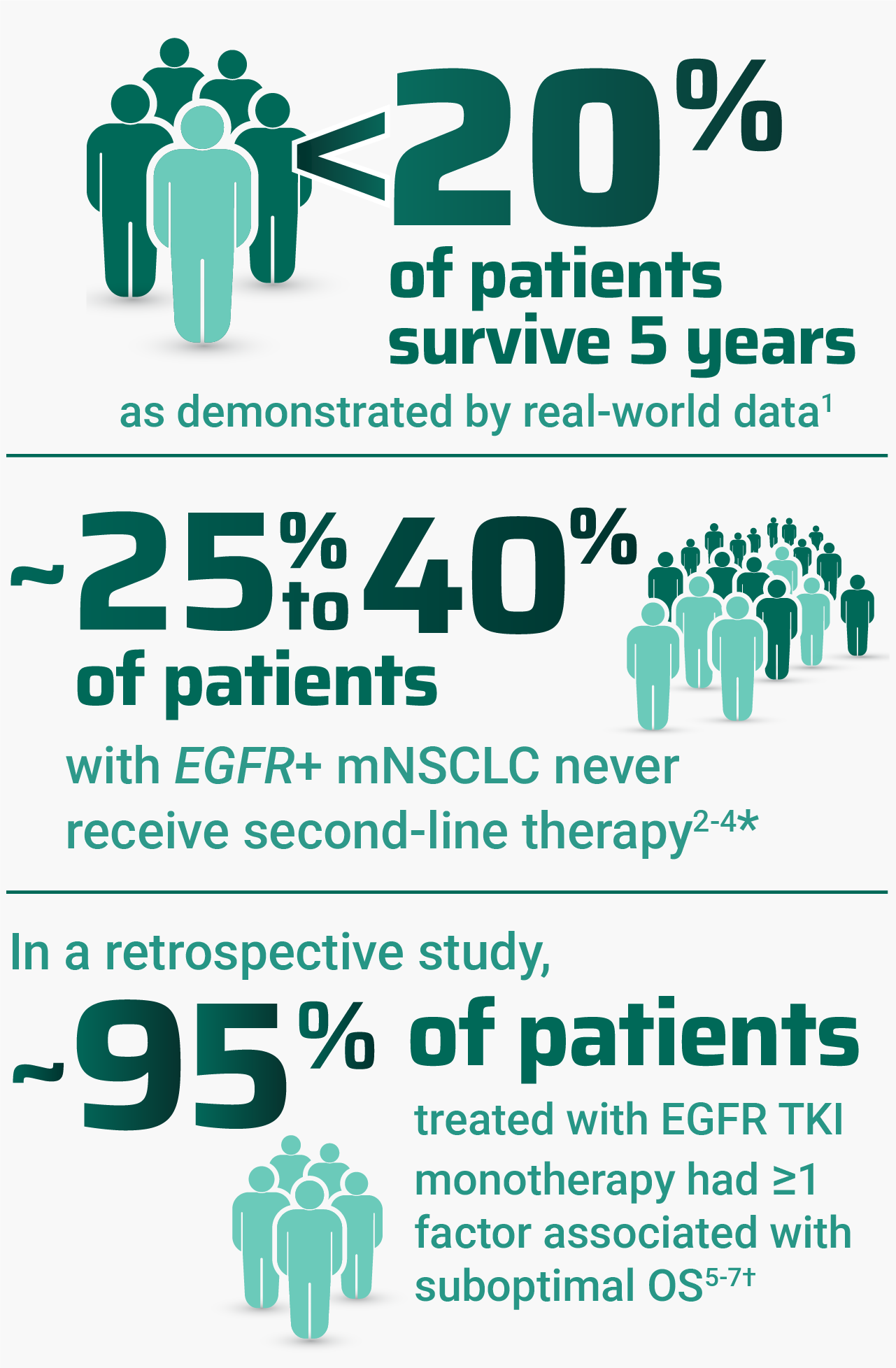
Despite advancements, there are many challenges associated with treating EGFR+ mNSCLC
Patient preference is another key factor in treatment decision-making
In a survey, patients ranked
extending their lives as the most important attribute of treatment8‡
In a separate survey, 70% to 90% of patients with advanced NSCLC
said they prefer chemotherapy-
free treatment when considering
first-line options9§
Regardless of treatment administered, patients with high-risk factors may experience poor outcomes, including OS. OS outcomes for patients with ≥1 risk factor in the MARIPOSA study are currently not available; thus, no conclusions can be drawn on RYBREVANT® (amivantamab-vmjw) and LAZCLUZE® (lazertinib) at this time.2,5,7,10
*Range includes patients who died or discontinued the assigned therapy without receiving second-line therapy during follow-up.2–4
†As demonstrated by Spira et al and Sabari et al, 2 retrospective cohort studies that used real-world electronic health record data from April 2018 to December 2023. Key individual factors associated with shorter real-world survival included: metastases of the liver, bone, and central nervous system; ECOG PS ≥2; age ≥65 years; and mutations of TP53, CDK-4, and EGFR L858R. The data sources reflected real-world clinical practice settings and actual data captured at the site of care, which may mean some data entries were missing or erroneous.5,6
‡Data come from a cross-sectional online survey of 160 adult patients, 30 care partners, and 150 clinicians conducted between March and May 2023. Survey questions were informed by a targeted literature review, qualitative interviews, and input from a steering committee that included 2 oncology physicians, an oncology nurse practitioner, a patient advocate, patient, and care partner. Descriptive statistics were generated and reported in aggregate. In addition to “extending their lives,” patients ranked “quality of life” and “functional independence” as the next most important treatment attributes.8
§Data come from a survey fielded from February to March 2025 of 150 US patients with stage IIIB, IIIC, and IV NSCLC.9
CDK-4, cyclin-dependent kinase 4; ECOG PS, Eastern Cooperative Oncology Group performance status; EGFR, epidermal growth factor receptor; mNSCLC, metastatic non–small cell lung cancer; NSCLC, non–small cell lung cancer; OS, overall survival; TKI, tyrosine kinase inhibitor; TP53, tumor protein p53.
EGFR Exon20ins
Exon 20 insertion mutations are the third most frequent EGFR mutations11
Testing with next-generation sequencing (NGS) at diagnosis is critical to detect EGFR mutations and select the most effective first-line treatment12,13
CF/CT DNA, cell-free/circulating tumor DNA; DNA, deoxyribonucleic acid; exon20ins, exon 20 insertion.
References:
- Bazhenova L, Minchom A, Viteri S, et al. Comparative clinical outcomes for patients with advanced NSCLC harboring EGFR exon 20 insertion mutations and common EGFR mutations. Lung Cancer. 2021;162:154-161. doi:10.1016/j.lungcan.2021.10.020
- Nieva J, Karia PS, Okhuoya P, et al. A real-world (rw) observational study of long-term survival (LTS) and treatment patterns after first-line (1L) osimertinib in patients (pts) with epidermal growth factor receptor (EGFR) mutation-positive (m) advanced non-small cell lung cancer (NSCLC) [ESMO abstract 1344P]. Ann Oncol. 2023;34(suppl 2):S774. doi:10.1016/j.annonc.2023.09.2377
- Lee JY, Mai V, Garcia M, et al. Treatment patterns and outcomes of first-line osimertinib-treated advanced EGFR mutated NSCLC patients: a real-world study. J Thorac Oncol. 2022;17(9)(suppl):S440. doi:10.1016/j.jtho.2022.07.764
- Girard N, Leighl NB, Ohe Y, et al. Mortality among EGFR-mutated advanced NSCLC patients after starting frontline osimertinib treatment: a real-world, US attrition analysis. J Thorac Oncol. 2023;18(4)(suppl):S51-S52. doi:10.1016/s1556-0864(23)00273-3
- Spira AI, Ran T, Lin I, et al. Risk factors associated with suboptimal real-world outcomes in patients with EGFR-mutated non-small cell lung cancer treated with front-line recommended therapy. Adv Ther. 2025;42(7):3547-3561. doi:10.1007/s12325-025-03234-3
- Sabari JK, Yu HA, Mahadevia P, et al. Overall survival after treatment with first-line osimertinib for EGFR-mutant advanced NSCLC in the US. Presented at: the World Conference on Lung Cancer; September 7-10, 2024; San Diego, CA, USA. E-poster.
- Felip E, Cho BC, Gutiérrez V, et al. Amivantamab plus lazertinib versus osimertinib in first-line EGFR-mutant advanced non-small cell lung cancer with biomarkers of high-risk disease: a secondary analysis from MARIPOSA. Ann Oncol. 2024;35(9):805-816. doi:10.1016/j.annonc.2024.05.541
- Orr LD, Vanderpoel J, Vadagam P, et al. Patient, care partner, and provider voice in treatment decision-making for non-small cell lung cancer. Patient Educ Couns. 2025;136:1-7. doi:10.1016/j.pec.2025.108776
- Data on file. Janssen Biotech, Inc.
- RYBREVANT FASPRO™ [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
- Riess JW, Gandara DR, Frampton GM, et al. Diverse EGFR exon 20 insertions and co-occurring molecular alterations identified by comprehensive genomic profiling of NSCLC. J Thorac Oncol. 2018;13(10):1560-1568. doi:10.1016/j.jtho.2018.06.019
- Pennell NA, Mutebi A, Zhou Z-Y, et al. Economic impact of next-generation sequencing versus single-gene testing to detect genomic alterations in metastatic non-small-cell lung cancer using a decision analytic model. JCO Precis Oncol. 2019;3:1-9. doi:10.1200/PO.18.00356
- Vanderpoel J, Stevens AL, Emond B, et al. Total cost of testing for genomic alterations associated with next-generation sequencing versus polymerase chain reaction testing strategies among patients with metastatic non–small cell lung cancer. J Med Econ. 2022;25(1):457-468. doi:10.1080/13696998.2022.2053403
- Leighl NB, Page RD, Raymond VM, et al. Clinical utility of comprehensive cell-free DNA analysis to identify genomic biomarkers in patients with newly diagnosed metastatic non–small cell lung cancer. Clin Cancer Res. 2019;25(15):4691-4700. doi:10.1158/1078-0432.CCR-19-0624
- Pennell NA, Arcila ME, Gandara DR, West H. Biomarker testing for patients with advanced non–small cell lung cancer: real-world issues and tough choices. Am Soc Clin Oncol Educ Book. 2019;39:531-542. doi:10.1200/EDBK_237863
- John A, Yang B, Shah R. Clinical impact of adherence to NCCN guidelines for biomarker testing and first-line treatment in advanced non-small cell lung cancer (aNSCLC) using real-world electronic health record data. Adv Ther. 2021;38(3):1552-1566. doi:10.1007/s12325-020-01617-2
- Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Non-Small Cell Lung Cancer V.2.2026. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed December 2, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.



